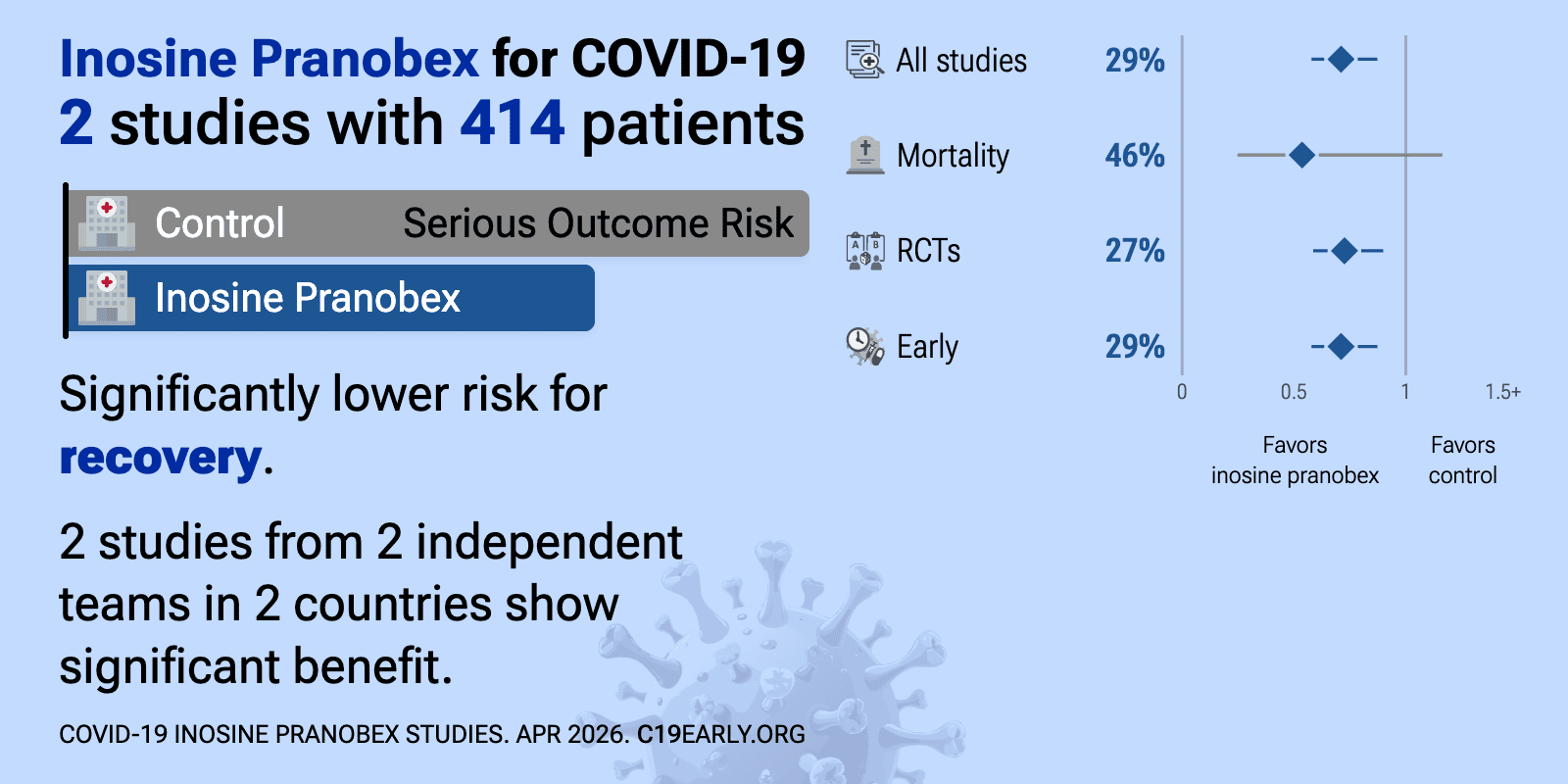
Inosine pranobex is an orally administered small-molecule immunomodulator with broad antiviral activity. It enhances natural killer cell cytotoxicity and T-lymphocyte function and promotes Th1-type cytokine responses, thereby augmenting host antiviral defenses.
Inosine Pranobex was adopted
in 1 country.
Recent:Jayanthi Veselý.
Apr 29 |
Meta-analysis of inosine pranobex studies | |
| Meta-analysis of inosine pranobex studies | ||
Mar 31 |
et al., International Journal of Infectious Diseases, doi:10.1016/j.ijid.2026.108519 | Inosine pranobex was associated with a reduction in mortality in COVID-19 by over 22%, as reported in a retrospective study utilizing complete health data from the Czech national registry |
| 46% lower mortality (p=0.11). Retrospective 67,841 inosine pranobex recipients matched 1:2 with 135,682 controls from the Czech national registry, showing significantly reduced COVID-19 mortality with treatment. Negative control outcome analyses (non-COVID mortality,.. | ||
Sep 16 2022 |
et al., Advanced Therapeutics, doi:10.1002/adtp.202200159 | Efficacy and Safety of Inosine Pranobex in COVID‐19 Patients: A Multicenter Phase 3 Randomized Double‐Blind, Placebo‐Controlled Trial |
| 27% improved recovery (p=0.003) and 33% greater improvement (p=0.0005). RCT 416 mild-to-moderate COVID-19 patients (both hospitalized and non-hospitalized) showing improved recovery with inosine pranobex. | ||